 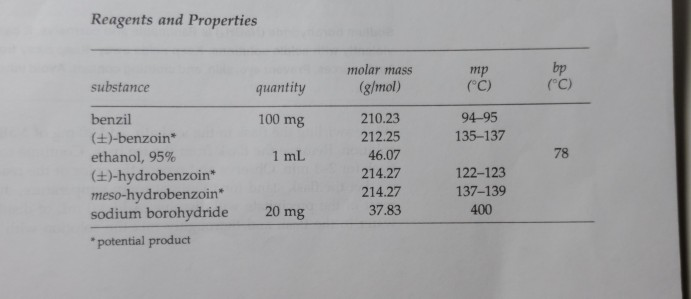
So, the overall formal charge of ethanol (CH3OH) is 0. Assuming the solution you used was 95 ethanol by volume and that the density of ethanol is 0.816g/mL, how much water and carbon dioxide are produced in the experiment Equation: C2H6O+3O2->2CO2+3H2O (You start off with 2.0 mL of ethanol) Can someone please help me I’m being timed and need help with 2 questions. So, the formal charge of each hydrogen is 0. Lets find out formal charge of Ethanol – CH3OH.įormal charge = Valence electrons – Unbonded electrons – 1/2 bonded electrons carbon gives tetrahedral molecular geometry.Oxygen gives bent molecular geometry whereas.Now, if you look out the entire molecular geometry, it gives two different results, such as, when you add 2 loan pair in hydrogen then 2 loan pair pushing down the hydrogen and as a result, it give bent molecular geometry. Oxygen – There are also 4 think attached to the oxygen.so, you can say that, it is a tetrahedral. In this work, we study the molar mass dependence of the thermodiffusion of polyethylene oxide at different temperatures in ethanol, water/ethanol mixture (c. it is not make bond and when add OH group then the bond angle will be 109.5°. The molecular masses of a carbon compound spread over a range of. Carbon – If you add few think on the center atom carbon then, when you add three hydrogen to the carbon, it sprayed out.Now, we can find out the molecular geometry of ethanol. According to the no of think attached to the center of carbon. Finding ethanols molar mass using vapor density Edition 1st Edition First Published 2015 Imprint CRC Press Pages 12 eBook ISBN 9780429089688. See also our theoretical yield calculator for chemical reactions (probably your next stop to finish the problem set). Do a quick conversion: 1 grams Ethanol 0.021706834440237 mole using the molecular weight calculator and the molar mass of CH3CH2OH. Hybridisation is the process in which two or more atomic orbitals with almost the same energy levels mix to form new hybrid orbitals. So, the 3 hydrogen and one OH group are attached in the center atom (carbon). The molar mass of C2H6O (ethanol) is: 46.069 grams/mol. Hence, one mole of Ethanol weighs (46.07) grams. There are few think are attached to the center atom (carbon). So, first focus on the carbon and then looking to the oxygen.įirst, look out the center atom (carbon). first term is carbon and other is oxygen. There are two main terms in the structure of ethanol is carbon and oxygen. Please refer to NLM’s Technical Bulletin for additional resources.The Lewis structure of ethanol is tell that it has electrons in the atom rang around the atom. National Oceanic and Atmospheric Administration’s CAMEO Chemicals ( ), available as a mobile application and downloadable software.The base peak in the mass spectrum of ethanol is at m / z 31. Ritz crackers Sodium chloride NaCl 58.4 4 ionic Soluble in water Slightly soluble in ethanol Boiling point is 1465 0 C Melting point is 800.7 0 C Used as a seasoni ng-maintains blood pressure-Too much salt. ethanol, also called ethyl alcohol, grain alcohol, or alcohol, a member of a class of organic compounds that are given the general name alcohols its molecular formula is C2H5OH. 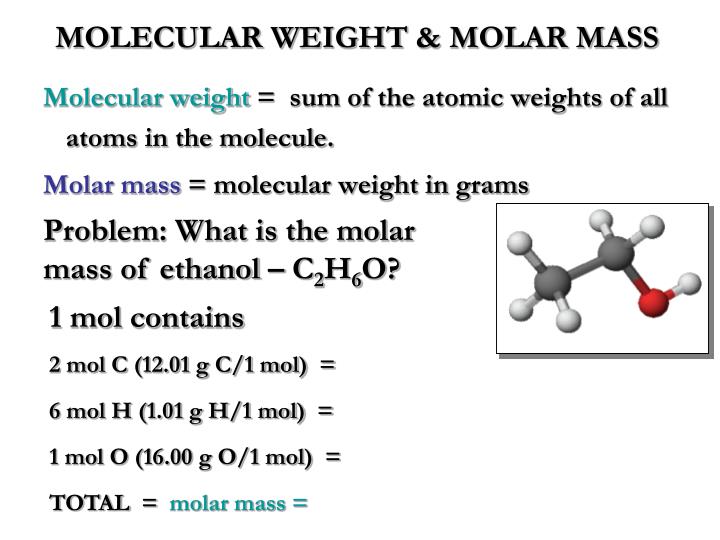 
Department of Transportation’s (DOT) Pipeline and Hazardous Materials Safety Administration Emergency Response Guidebook ( ), available as a downloadable document and a mobile application Thus, using high resolution mass spectrometer one can choose the correct structure out of. Chemical Compounds Molar Mass Research Project Objectives: To identify the various ionic and molecular compounds found in processed food. Standard molar entropy, S o liquid: 159.9 J/(mol K) Enthalpy of combustion, c H o: 1370.7 kJ/mol Heat capacity, c p: 112.4 J/(mol K) Gas properties Std enthalpy change of formation, f H o gas: 235.3 kJ/mol Standard molar entropy, S o gas: 283 J/(mol K) Heat capacity, c p: 78.28 J/(mol K) at 90 C 87. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
